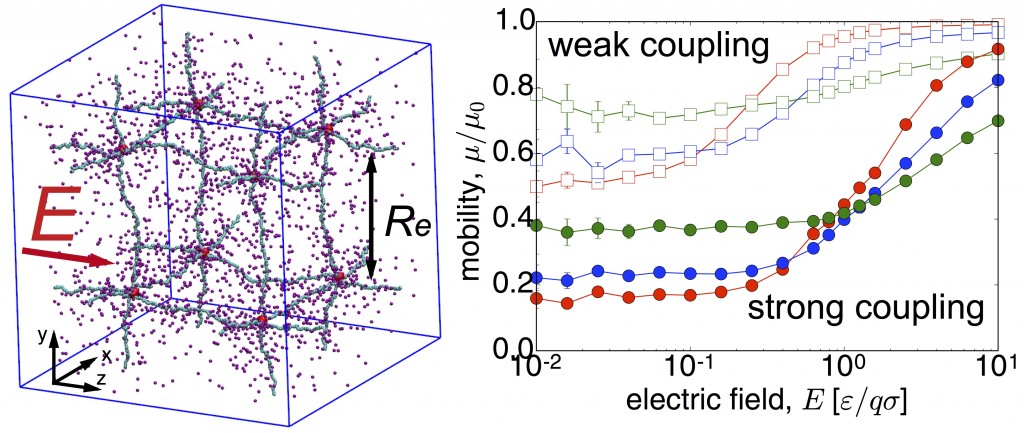
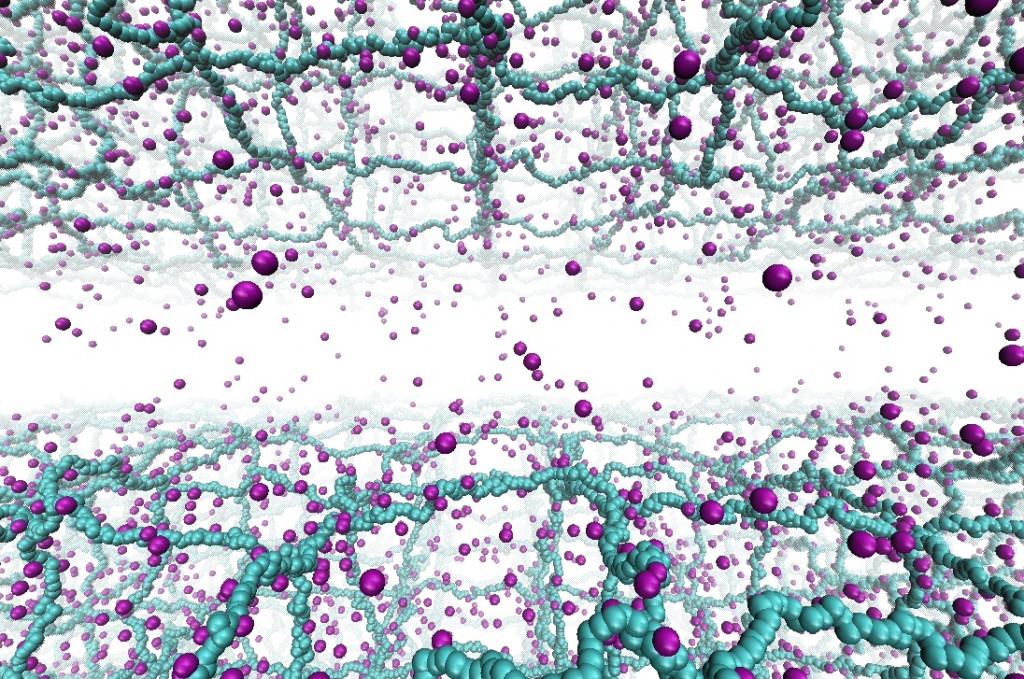
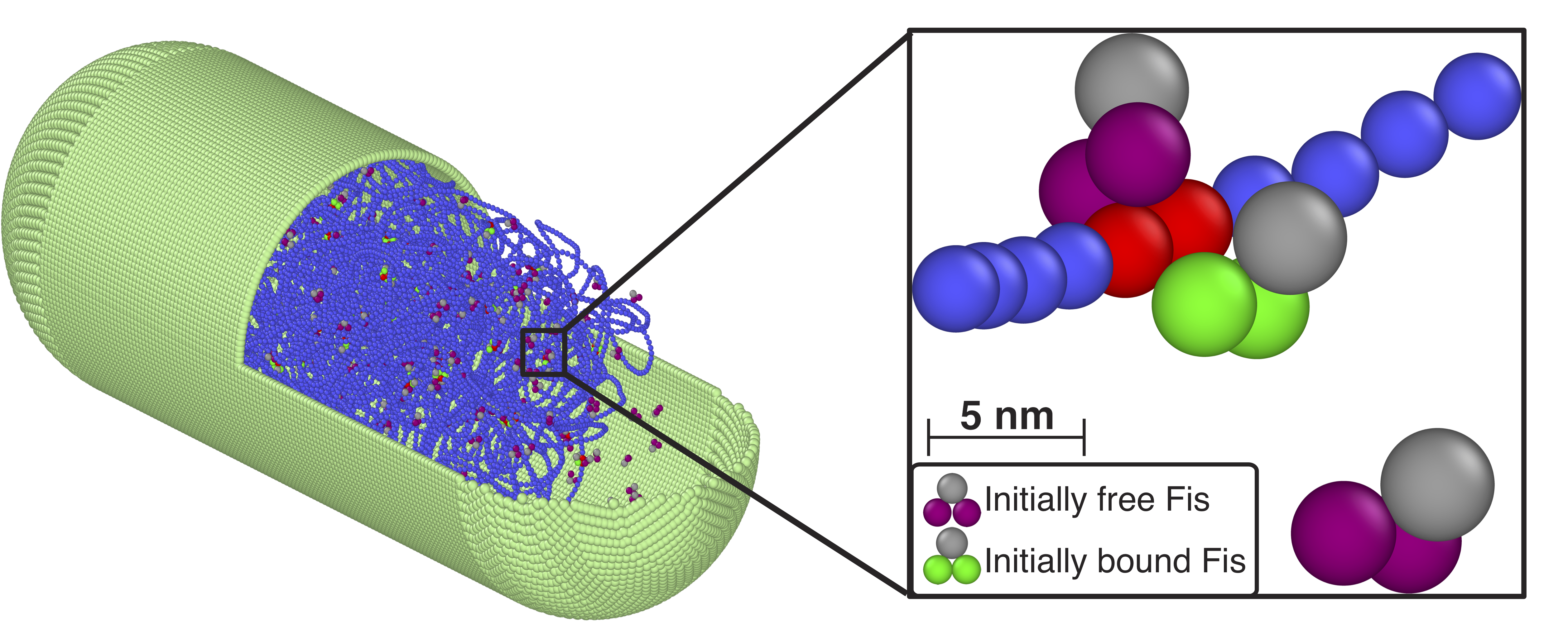
Concentration-dependent off-rates of DNA-binding proteins in bacterial confinement
Almost all cellular functions from replication to transcription are controlled by the transient interactions between DNA and DNA-binding proteins. By using MD simulations of large-scale bacterial model, in which long DNA chain can interact with dimeric transcription factor proteins such as Fis or HU, we show that protein concentration influences the off-rates of the DNA-bound proteins. These proteins can also structurally change the chromosome compactions and further alter the off-rates by creating DNA-protein complexes and highly-collapsed regions of DNA. Read more on Biophysical J.
 |
 |
Inhomogeneous chromatin flexibility contributes to the nuclear organization in eukaryotic cells
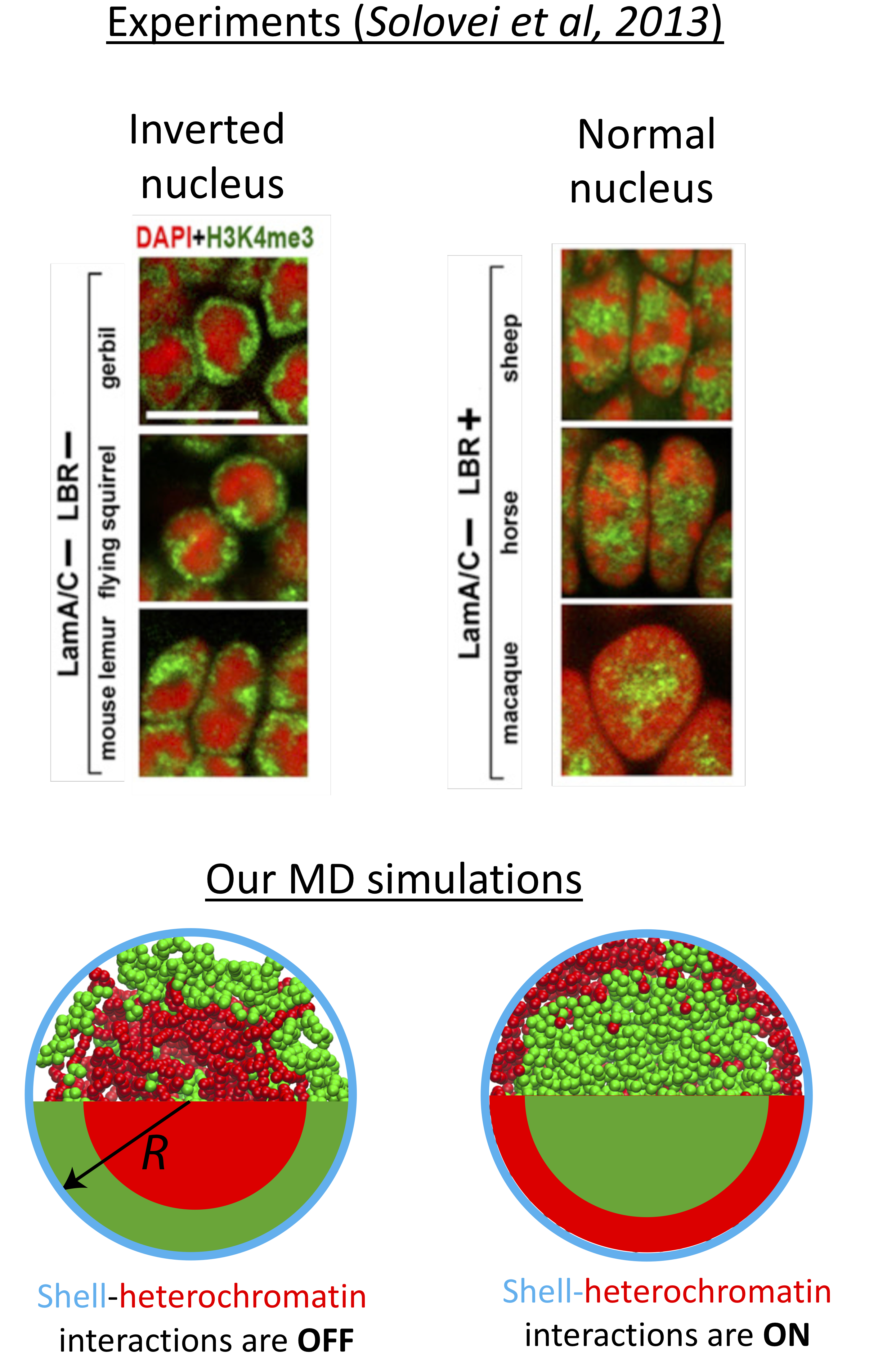
When we look into the organization of genome in the confinement of the cell nucleus, first thing we notice is that distribution of chromatin (a fibrous structure composed of protein-DNA complexes) is not random. In most healthy cells, while gene-passive heterochromatin (red regions in the images above) is found near the interior surfaces of the nuclear envelope, gene-active euchromatin (green) localizes in the nuclear interior. Yet, deviations from regular chromatin distribution are frequent, for example, upon aging, under malignant diseases, or even naturally in rod cells of nocturnal mammals, for instance, when proteins connecting the chromatin to the nuclear lamine is deficient or absent. In order to understand how polymer properties of the long chromatin fiber influences the genome organization, we study the segregation of heterochromatin in cell nucleus by modeling interphase chromosome as diblock ring copolymers confined in a rigid spherical shell by using extensive Molecular Dynamic simulations. Heterochromatin block of each bead-spring chain mildly differs from that of euchromatin via its bending stiffness. Our simulations indicate that in the absence of attractive interactions between nuclear shell and chromatin, the majority of heterochromatin segregates in the nuclear interior due to depletion of less flexible heterochromatin segments from nuclear periphery. The central coalescence of heterochromatin is in accord with experimentally observed positional inversion of heterochromatin in rod cells and exhibits a maximum when heterochromatin is only a factor of two less flexible than euchromatin. Attractive interactions between heterochromatin segments and the bond-crossing (topoisomerase activity) further increase the heterochromatin inversion. The inversion is also found to be independent of the heterochromatin concentration and chromosome number. The inverted nuclei can be reversed by switching on the attraction interactions between the nuclear shell and heterochromatin or by increasing the bending stiffness of the heterochromain. Overall, our results indicate that any molecular phenomenon that can lead to suppressed bending fluctuations along the chromatin could favor nuclear segregation, thus, can have important consequences for gene regulation. Read our preprint.
How receptor density and reservoir dimensions effect the diffusion process of ligands initially located at their receptors
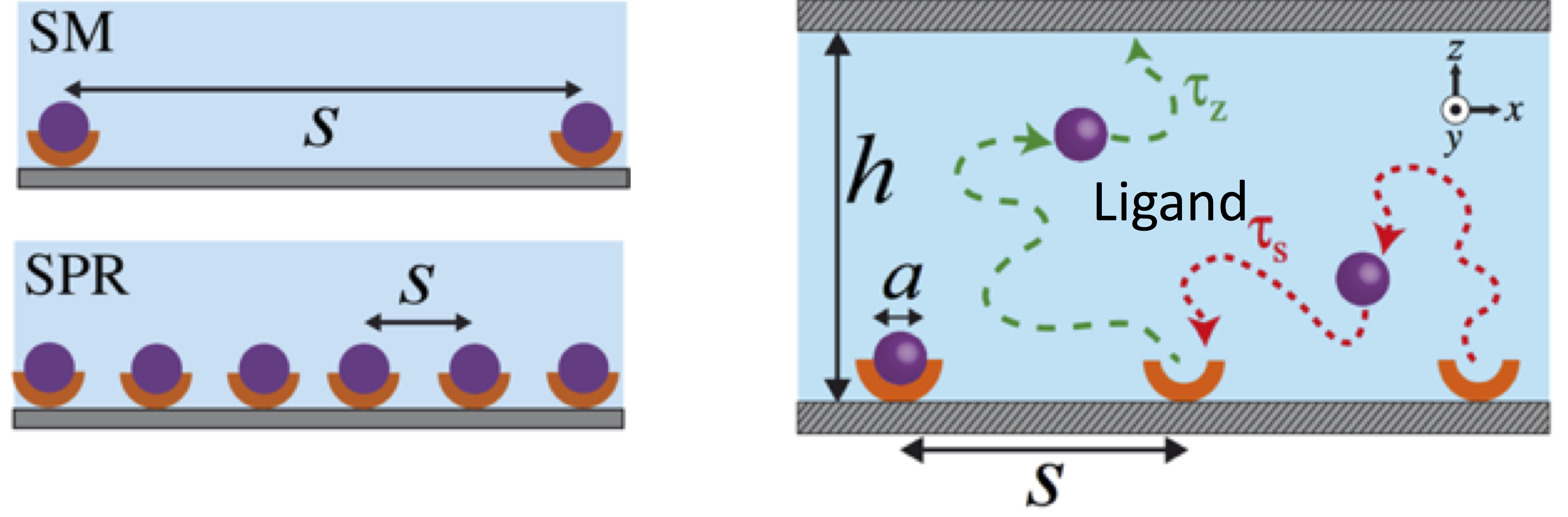
Single-molecule (SM) or SPR (Surface Plasmon Resonance) experiments rely on relaxation of concentration quenches of initially surface-bound molecules into confined reservoirs to determine molecular kinetic rates. Similarly, biological processes such exocytosis, in which small molecules are emitted into the intracellular cleft for cellular communication, can be considered to be a relaxation process of an effective concentration quench. We study a model system closely related to the above cases in which weakly interacting Brownian particles are released from their binding sites into a confined volume by using molecular dynamics simulations and scaling arguments. Our results suggest that the rebinding rate of released particles exhibits various power laws until the confined volume is entirely filled by the particles. Furthermore, the cumulative rebinding rate, which is time integration of the rebinding rate, exhibits a novel plateau behaviour. This plateau is a result of the minimal number of collisions between the binding sites and ligands. Our results can have important consequences for molecular signalling as well as for the interpretation of kinetic measurements of ligand-receptor interactions. Read more on Biophysical Journal.
Protein-DNA interactions and their role in the 3D structure of the genome
Inside the nucleus of eukaryotic cells (e.g., mammalian cells), the meters-long DNA molecule is found in various compaction states to be able to fill micro-meter size nuclear volumes. In some cases, a more compact form of the DNA (i.e., heterochromatin) resides near the interfaces of the cell nucleus, whereas, in other nuclear locations, we observe a less compact form of the DNA (i.e., euchromatin). The DNA can fold these special forms as a result of its interactions with a vast array of heterogeneously distributed nucleic acid binding proteins and ions (monovalent or multivalent salts such as Na+, Mg2+). Strikingly, any irregularity in DNA compaction states can be traced back to DNA-protein interactions, which are also hallmark of malignant disorders, including cancer and progeria (i.e., early aging) syndrome. In our research, by considering the polymer nature of the negatively charged DNA molecule, we would like to quantitatively model the “phase” segregation of DNA by borrowing arguments from physics of polymers and self-assembly. Our ultimate goal is to decipher the fundamental mechanism of DNA organisation in confinement, thus, resolve unknowns of mechanochemical aspects of diseases mechanisms.

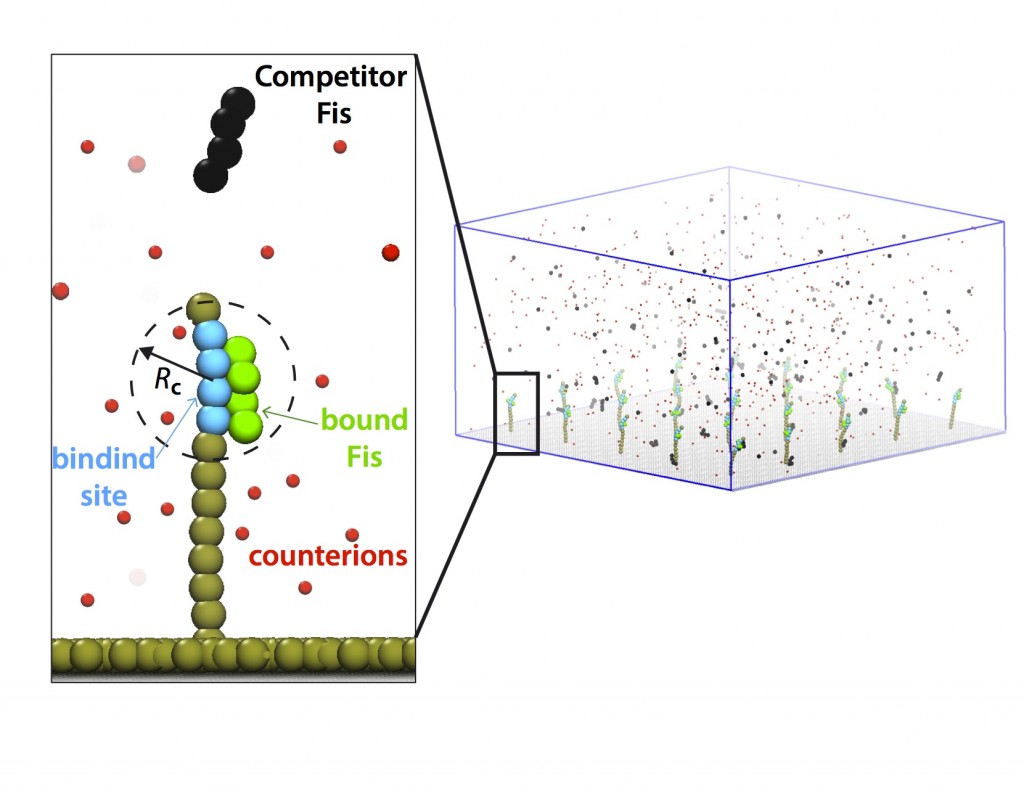
The standard picture of biomolecular interactions posits that the residence time (off rate) of transcription factors (TFs) on DNA molecules in cells is independent of their concentration in solution. We designed and performed single-molecule experiments and simulations, where we measured dissociation kinetics of Fis (factor for inversion stimulator) – a key TF in E. coli bacteria and also major bacterial nucleoid protein — from single dsDNA binding sites. We find that the residence times (i.e., inverse off-rates of disassociation events) saturate at large protein concentrations in solution. While spontaneous (i.e., no unbound proteins in solution) dissociation shows a strong salt dependence, the facilitated dissociation depends only weakly on salt. A theoretical model quantitatively explain our findings successfully and show the prevalence of this phenomenon. Read more on PNAS.
Polyelectrolyte Hydrogels
Hydrogels are highly swollen networks of charged polymers. Apart, from being the main constituent of contact lenses, pill capsules, etc., they have also proven themselves pretty effective in other applications, such as mechano-electric energy converters, electrolyte materials, energy storage or for bio-mimetic hybrid, structures.
- Electrostatic energy conversion with polyelectrolyte gels
 Hydrogels swell due to the balance between osmotic pressure of ions and elasticity of the polymer network. The balance between these inherent electrostatic and mechanic properties of hydrogels allows them to adjust their internal electrostatic interactions reversibly upon application of external electromagnetic fields or mechanical deformations. Our MD simulations with explicit counterions suggest that if a hydrogel undergoes strain-control deformations, an amount of mechanical energy applied is converted into electrostatic energy rather than elastic and excluded volume energies. The energetic change is due to the decreasing translational entropy of counterion gas trapped inside the gel in combination with polymeric features of PE gels. This scenario is only possible if ionized counterions cannot vacate the gel, which is ensured by the electroneutrality condition inside the gel. Read more on ACS Macro Letters.
Hydrogels swell due to the balance between osmotic pressure of ions and elasticity of the polymer network. The balance between these inherent electrostatic and mechanic properties of hydrogels allows them to adjust their internal electrostatic interactions reversibly upon application of external electromagnetic fields or mechanical deformations. Our MD simulations with explicit counterions suggest that if a hydrogel undergoes strain-control deformations, an amount of mechanical energy applied is converted into electrostatic energy rather than elastic and excluded volume energies. The energetic change is due to the decreasing translational entropy of counterion gas trapped inside the gel in combination with polymeric features of PE gels. This scenario is only possible if ionized counterions cannot vacate the gel, which is ensured by the electroneutrality condition inside the gel. Read more on ACS Macro Letters.
- Ionic DC conduction in polyelectrolyte hydrogels
Ionic transport in heterogeneous media on the microscale and nanoscale is of paramount interest for many applications including water desalination, biomimetic systems, as well as the potential for future generations of man-made devices, which can interface and be intrinsically compatible with biological tissues. Coarse-grained molecular dynamics simulations suggest that a nonlinear response of the ionic conductivity to an applied electric field, for field strengths that are comparable to the ionic coupling strengths. This behavior correlates to a broadening of the ionic distribution around the polymer backbone under an increasing electric field. Also, we find that the weak-field ionic mobility in gels increases with density, which is opposite to the behavior of simple electrolytes. This relates to the mean coupling between charges that decreases in gels, but increases in simple electrolytes, with increasing density. These results provide more insights into the electric response of polyelectrolyte gels, to support the development of applications that combines electric and mechanical properties of polyelectrolyte gels for energy storage, sensing, selective transport, and signal transfer. Read more on Macromolecues .
- Interactions between charged polymeric surfaces
Hydrogels are highly-swollen networks of cross-linked polymers with ionizable groups. Surfaces formed by these charged-polymers (polyelectrolytes) are highly abundant in both synthetic and biological systems. These charged-polymer networks are the main constituents of contact lenses and adsorbing components of diapers, and also appear in various pharmaceutical applications in the forms of smart drugs due to their pH response. In biological machinery, hydrogels composed of biopolymers appear quite often as well, particularly in connective and inter-cellular tissues: cartilage tissue and its components (synovial joints), mucin bilayers bridging the corneal stroma and epithelial surfaces in eye, mucin-brush layers coating the interior surfaces of mammalian respiratory system (lungs) are among some examples. It is, hence, desirable to understand the interactions between the hydrogel surfaces and possibly relate experimentally measurable properties (e.g., swelling ratio or lubrication properties) to the molecular details. In our recent publication, we first revisit a previous theory based on the solution of the Poisson-Boltzmann equation and compare the results to our molecular dynamics simulations of two highly-charged hydrogel surfaces. We also introduce scaling arguments to relate electrostatic strength of solvent (also referred to as ionic strength) and polymerization degree to the observed pressure regimes and the threshold pressure separating these regimes. Read more on Macromolecules.
Self-assembly of Amphiphilic Molecules
- Co-Assembly of Peptide Amphiphiles (PAs) and Lipids into Supramolecular Nanostructures
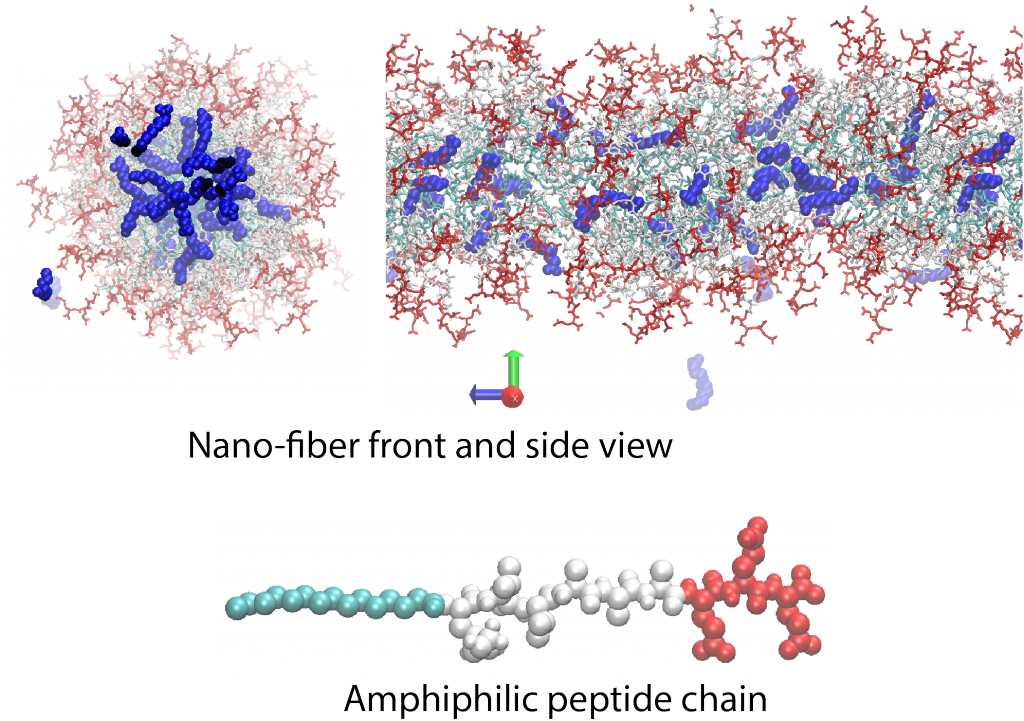
An amphiphilious peptide molecule is composed of hydrophilic (red) and hydrophobic (cyan and white) building blocks. These molecules can self-assemble and form 2D and 3D structures, such as micron-size nano-fibers, micelles or bilayers depending on solution pH, hydrogen bonding
capacity of peptides (sequence) or salt concentration. Non-covalent nature of these structures, as well as their amino-acid building blocks, highlight them for pharmaceutical applications where bio-degradability is paramount. We investigate the formation of these nanostructures by using atomistic molecular dynamics simulations and analytical theories.
Co-assembly of binary systems driven by specific non-covalent interactions can greatly expand the structural and functional space of supramolecular nanostructures. Examples include co-assembly of bioactive and nonbioactive molecules to modify the intensity of smart drugs, or functional biomimetic structures. In our work, the self-assembly of peptide amphiphiles (PAs) and fatty acids (lipids) driven primarily by anion−π interactions are focused. With a low content of the lipid, the cylindrical nanofiber morphology is preserved. However, as the aromatic units are placed along the peptide backbone away from the hydrophobic residues, the interactions with the lipids transform the cylindrical supramolecular morphology into ribbon-like structures. Increasing the stoichiometric ratio of the lipid to PA leads to either the formation of large spherical vesicles in the binary systems or a heterogeneous mixture of assemblies. Our findings reveal that specific inter-molecular interactions can drastically change supramolecular morphology and bridge between nano to micro scales. Read more on our work at JACS.
- Formation of ion-rich phases in the melts of amphiphilic molecules.
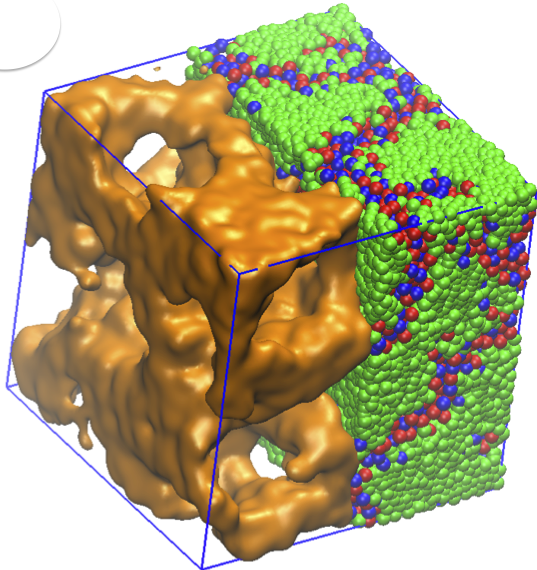 Amphiphilic molecules bear both hydrophobic and hydrophilic chemical groups. At high volume fractions, these molecules arrange themselves relative to each other in order to minimize their free energy and form phases. Ionic liquids – with aliphatic tails and cationic groups – exhibit fascinating nanoscale morphological phases and are promising materials for energy storage applications. Liquid crystalline order emerges in ionic liquids with specific chemical structures. In our study, we investigate the phase behavior and related ionic conductivities of dry (solvent free) ionic liquids, using extensive molecular dynamics simulations. Temperature dependence, properties of polymeric tail and excluded volume symmetry of the amphiphilic ionic liquid molecules are investigated in large-scale systems with both short and long-range Coulomb interactions. Our results suggest that by adjusting stiffness and steric interactions of the amphiphilic molecules, lamellar or 3D continuous phases result in these molecular salts. We also demonstrate that, in the absence of electrostatic interactions, the morphology is distorted. Our findings inspire new design principles for room temperature ionic liquids and help explain previously-reported experimental data. Read more on PCCP .
Amphiphilic molecules bear both hydrophobic and hydrophilic chemical groups. At high volume fractions, these molecules arrange themselves relative to each other in order to minimize their free energy and form phases. Ionic liquids – with aliphatic tails and cationic groups – exhibit fascinating nanoscale morphological phases and are promising materials for energy storage applications. Liquid crystalline order emerges in ionic liquids with specific chemical structures. In our study, we investigate the phase behavior and related ionic conductivities of dry (solvent free) ionic liquids, using extensive molecular dynamics simulations. Temperature dependence, properties of polymeric tail and excluded volume symmetry of the amphiphilic ionic liquid molecules are investigated in large-scale systems with both short and long-range Coulomb interactions. Our results suggest that by adjusting stiffness and steric interactions of the amphiphilic molecules, lamellar or 3D continuous phases result in these molecular salts. We also demonstrate that, in the absence of electrostatic interactions, the morphology is distorted. Our findings inspire new design principles for room temperature ionic liquids and help explain previously-reported experimental data. Read more on PCCP .
Friction of Soft Matter at Molecular Scales
- Polymer-brush friction
Polymer-brush systems have been attracting scientists for the last two decades extensively. On the other hand, polyelectrolytes, which are polymer chains of charged monomers, have unique properties and are quite abundant in biological systems; DNA and most proteins can be counted among many. Experiments and simulations have shown that the friction coefficient between two polyelectrolyte brushes is less than 10^-4, which make them an excellent candidate for effective build-in lubricants. We would like to use coarse-grained computer models to understand and invoke further effects of other factors such as salt, velocity, etc. on polyelectrolyte brush friction. Our computer models can also be used to check various scaling arguments quantitatively as well as quantitatively. Furthermore, since polyelectrolyte brushes carpet the surface of the cilia, our simulations can shed light on cilia-mucus systems as well.
- Friction in biological systems
In many daily-life engineering applications, friction is a drawback to be minimized for efficiency. Conversely, in biological systems involving self-assembled building blocks held together by non-covalent interactions, friction is necessary to maintain the biological machinery. For instance, it is the friction that determines the ligand-receptor interactions or the folding times of proteins together with their free energy landscape. Similarly, for iso-energetic processes, for which the free energy barrier is either absent or low, such as a kinesin motor sliding on a microtubulin or a macromolecule diffusing on a surface, the friction manifests itself in the corresponding diffusion coefficient. To characterize the friction in such biological processes is more challenging compared to macro-solid systems, such as two blocks sliding relative to each other, where a single friction constant quantifies the friction. Read more here.
However, for biological soft systems, friction depends on the conformations of the molecules since these conformational changes affect the intra and inter-molecular interactions. For example, in a protein folding process, where the folding is preceded by an initial collapse of the amino acid chain, the number of native contacts increases and the water contact decreases as the folded state is approached. Not surprisingly, the confinement level of monomers by other monomers increases towards the folded state, and so does the friction. Read more here.
Air-Surface layers: Two main components of clearance mechanisms in the human respiratory system are viscoelastic mucus and cilia. The mucus layer driven by antiplectic beats of cilia not only cleans the respiratory tract system but also protects the epithelial layer from external factors such as viruses. Our aim is to combine concepts of polymer physics and hydrodynamics to build an understanding of the mucus-cilia machinery. Such a minimal model, which can reproduce in vivo as well as in vitro experimental results, can draw boundaries between healthy and diseased systems and quantify the mucus-cilia interactions in more detail.